
This gives rise to high electrical and thermal conductivities as well as high optical reflectivities. What distinguishes metals from all other solids is the ability of the electrons to readily respond to applied electric fields, thermal gradients, and incident light. Even though individual electron–electron interactions are repulsive, there is sufficient electrostatic attraction between the free-electron gas and the positive ion cores to cause bonding. In metals the outer valence electrons of each atom form part of a collective free-electron cloud or gas that permeates the entire lattice. The so-called metallic bond occurs in metals and alloys. Milton Ohring, in Materials Science of Thin Films (Second Edition), 2002 1.4.3.1 METALLIC 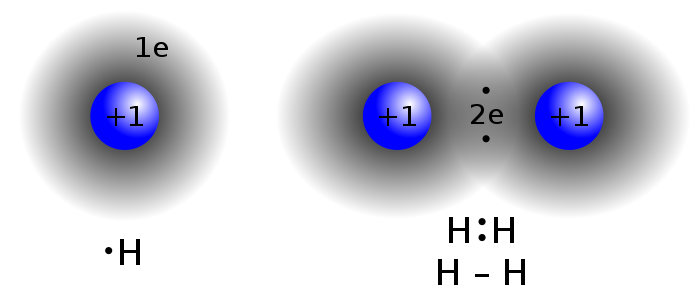
Depending on the application, the testing of thermal shock resistance, creep behaviour and oxidation resistance are further possible examinations of metal–ceramic compounds. The examination is usually done on a real component using a leak detector (e.g. in power electronics, an examination of the gas tightness of the brazed seam is of great interest. In addition, ceramics are known to exhibit only a low tolerance to tensile stresses. 16 The tensile test, which is widely used for testing metallic materials, is rarely applied in testing metal–ceramic compounds because the treatment of the ceramic for fixing is very problematic. But even here the transferability of test results is complicated due to the geometry dependence. Here, two butt-brazed sample plates are sheared off in a special test device. 16įurthermore, strength can be assessed by a shear test. The determined data can only be used for comparative purposes. For this reason, transferability of values is difficult. Also, the geometry of the test specimens should be given, since the strength values are geometry dependent. Therefore, when specifying strength values, it is crucial to ndicate whether the three- or the four-point bending test is used. Due to the larger stressed volume, the failure probability for the four- point bending test is higher, and lower strength values are determined. 
The bending test with three-point bending measures a higher strength than the four-point bending test, because the influence of the volume on the strength is of considerable importance for ceramic materials. 16 Weibull diagrams and parameters, which are standard in ceramics research, 38 can be used to evaluate the strength values to base the measured values on a statistic. Because of the usual high scattering of strength values in ceramic materials, bending tests of metal–ceramic compounds also lead to very high scattering values of up to ± 50% compared with the mean. 
Wielage, in Advances in Brazing, 2013 12.7 Examination methods for brazed metal–ceramic compoundsĮxamination methods for brazed metal–ceramic compounds include strength tests (mostly bending tests) and optical and scanning electron microscope studies. With increasing localization of valence electrons, covalent interactions cause deviations from spherically symmetric bonding, leading to more complicated structures. The metallic bond in typical metals is non-directional, favoring structures corresponding to closest packings of spheres. Quantum-mechanical calculations show that in large agglomerations of metal atoms the delocalized bonding electrons occupy lower energy levels than in the free atoms this would not be true for isolated “metal molecules”. Thus, the well-defined electron localization in bonds connecting pairs of atoms with each other loses its meaning. In typical metals, like the alkali metals, these outer orbitals are spherical s-orbitals allowing overlapping with up to 12 further s-orbitals of the surrounding atoms. The main difference between these two bond types is that the ionization energy for electrons occupying the outer orbitals of the metallic elements is much smaller. The metallic bond can be described in a similar way as the covalent bond. STEURER, in Physical Metallurgy (Fourth Edition), 1996 2.1.2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
